UDC 621.039:548.3
Effects of Fuel Rod Cladding Temperature and Stressed Conditions on Hydride Reorientation
T.P. Chernyayeva, V.M. Grytsyna, V.S. Krasnorutskyy, A.P. Riedkina, I.A. Petelguzov, Ye.A. Slabospitskaya
‘Nuclear Fuel Cycle’ Science and Technology Establishment National Science Center “Kharkov Institute of Physics and Technology” Kharkov, Ukraine
gritsina@kipt.kharkov.ua, тел.+3 8 057 335 67 81
Abstract
This paper presents results of experimental research into hydride reorientation and hydrogen embrittlement, which may occur in the SNF FR cladding at conditions simulating normal and some accident modes of SNF handling. We performed simulation experiments with examination of how SNF overheat to 450 ºC and stressed condition during loading into a SNF dry storage facility affect hydride reorientation at different hydrogen concentrations in the dummy claddings. Research was done to study changes in the hydride morphology during hydride reorientation tests at conditions simulating the handling operations of SNF loading into and sealing in storage baskets, and accident temperature increase in the SNF dry storage facility during SNF storage. It was established that at hydrogen concentrations in excess of 170 ppm, hydrogen reorientation in the dummy claddings during thermal tests begins at tensile stresses exceeding 55-60 MPa. For hydrogen concentrations in the claddings above 250 ppm, hydride reorientations begin at lower tensile stresses. In the dummy claddings at at 450 C ≥70 MPa and hydrogen concentration above 250 ppm, hydrogen reorientation occurs and the hydride reorientation factor increases significantly. Thermal cycling at the stage of SNF holding in the SNF dry storage facility significantly intensifies hydride reorientation. In the claddings at at 450 C=70 MPa and hydrogen concentration of 400 ppm, thermally cycled three times in the temperature range of 180 C450 C, almost complete hydride reorientation occurs.
Mechanical tests of samples with various hydrogen concentrations как before так and after the hydride reorientation tests were performed.
The results obtained and the information search conducted allow a prediction on the risk margin due to SNF cladding degradation related to the phenomena induced by presence of hydrogen accumulated in the Zr-1%Nb alloy FR cladding during operation.
Introduction
The management and long-term storage of spent nuclear fuel is an integral part of the nuclear fuel cycle. The safety of spent nuclear fuel handling operations largely depends on the SNF cladding condition. The SNF dry storage facility commissioned on August 24, 2001 at ZNPP (Zaporozhye NPP) site has considerably raised the demand for research into the phenomena responsible for the FR cladding degradation during SNF handling and long-term storage.
For many years Zirconium-base alloys has been used for the cladding of the fuel rods and structural components of fuel assemblies in water cooled power reactors, like pressurized water reactors (PWRs, VVER), boiling water reactors (BWRs, RBMK) and pressurized heavy reactors (HPWRs) [1] Upon completion of their operation, fuel rods remain internally pressurized [2, 3] and retain a number of degrading factors, such as hydrogen, accumulated during operation [4].
Degradation phenomena induced by presence of hydrogen in zirconium include:
— hydrogen embrittlement (HE), which is a significant decrease in ductility caused by hydrogen uptake [5, 6];
— delayed hydrogen cracking (DHC), which is gradual crack growth caused by discrete cracking along the hydrides formed at its tip [7, 8, 9];
— hydrogen redistribution and hydride blistering (formation of large hydrides in the local hydrogen accumulation areas) [10].
It is commonly believed that spent nuclear fuel (SNF) handling is mostly jeopardized by hydride reorientation and increased hydrogen embrittlement efficiency caused thereby [11].
Traditionally, the cladding tube manufacturing predetermines their proneness to formation of circumferentially (tangentially) oriented hydrides. The potential for hydride embrittlement of cladding fuel rods may increase drastically if the hydride platelets are reoriented from their normal circumferential to the radial direction of the cladding [12, 13, 14]. Hydride reorientation can occur as a result heating during the vacuum drying process in which the cladding temperature can reach up to 450 C, causing the hydrides to partially or completely dissolve. During subsequent cool-down, the hydrogen in solid solution can re-precipitate as radial hydrides if the hoop stress caused by the end-of-life internal pressure of the tangential stress exceeds the threshold stress level for the formation of radial hydrides [15].
By the end-of-life, the fuel rod internal pressure under the cladding, depending on the initial pressure (2.0…3.45 MPa), in the PWR rods at a room temperature is 4 …6 MPa [2, 16]. The dependence of the end-of-line pressure versus burnup (Bu) can be stated as follows

where Pend are internal fuel rod pressure at end of life; Bu is burnup in MWD/kgU, 2.8781 – mean initial pressure under the shell (fitting parameter).
According to US NRC requirements, the following criteria shall be met to guarantee fuel rod integrity during SNF handling and long-term storage [16, 17]:
— tangential stress in the fuel rod cladding at 400 C should not exceed 90 MPa;
— hydrogen concentration in the fuel rod cladding should not exceed 400-500 ppm.
The amount of hydrogen accumulated in Zr-1%Nb alloy during six years of operation (70 MWD/kgU) is 50…60 ppm and does not exceed 80 ppm; hydrides up to 80 microns long predominantly are tangentially oriented [18]. Due to such low hydrogen concentration, no limit was set for hydrogen uptake previously, however at present a design limit of 400 ppm has been implemented to match U.S. and western design criteria [17, 19].
This paper describes the hydrogen reorientation tests simulating the temperature mode and stress condition of Zr-1%Nb dummy claddings with hydrogen concentration up to 400 ppm and different scenarios of SNF handling and storage.
Experimental Procedure
Sample
The cladding used in the present study was non-irradiated Zr-1%Nb cladding tubes in the delivery condition (finish annealing 580 C, 3 hours); their main parameters are shown in Table 1.
Table 1 – Main Parameters of the Cladding Tubes Used
| Material | Chemical composition | Condition | Grain size, microns | Texture | Dimensions, mm | |
| Alloy E110 | Zr-1 wt.%Nb | Recrystallized | 3-5 | fR=0.586
fT=0.364 fL=0.050 |
Outer diameter – 9.13;
Wall thickness – 0.68 |
|
Treatment for Hydride Re-orientation
Hydride reorientation tests were performed on internally pressurized dummies with Zr-1%Nb cladding (Fig. 1). The overall dummy length was 112 mm, with the gas-filled cavity length of 66 mm. The gas (helium) pressure under the cladding was from 3 to 5 MPa.

Fig. 1 – Pressurized Dummy for the Hydride Reorientation Test
According to the specification, hydrogen concentration in non-hydrogenated Zr-1%Nb (E110) cladding tubes does not exceed 15 ppm (ppm –part per million); its standard concentration varies within 4-7 ppm [20]. The dummy claddings were saturated with hydrogen using a “dry” hydrogenation method at 380 and 420 C. Titanium hydride powder was used as the source of gaseous hydrogen. The hydrogen concentration was determined by weighing and using metallographic structure images (transverse cross-section). The hydrogen concentration in the cladding of the hydrogenated dummies ranged from 60 to 400 ppm.
The test temperature mode was based on SNF handling safety criteria.
The currently assumed safety criteria system of the Spent Nuclear Fuel Dry Storage at Zaporozhye NPP (ZNPP) meets the following [21]:
— the fuel rod peak cladding temperature operated in the fuel assemblies for the normal long-term dry storage in the helium environment is 350 C;
— for extreme weather conditions and during SNF handling the temperature may rise to 450 C for not more than 8 hours during the entire handling and storage period.
Changes in the cladding temperature of SNF fuel rods with 1 kW decay heat for long-term storage in the ZNPP SNF Dry Storage are shown in Fig. 2. The peak fuel rod clad temperature at the beginning is 349C (Fig. 3) and decreases with time. In 5 years this temperature will equal 275 C, and in 45-50 years it will reduce to 180 C.
 |
 |
| Fig. 2 – Changes in Fuel Rod Clad Temperature during Storage in ZNPP SNF Dry Storage | Fig. 3 – Temperature Mode during Tests Simulating Handling (Mode 1) and Tangential Stresses in the Dummy Cladding during Tests (P293 =3, 4 and 5 MPa) |
The temperature mode during tests simulating handling conditions (Mode 1) is shown in Fig. 3.
The stressed condition of both SNF fuel rod claddings in long-term storage and dummy claddings during tests is described by the following equations:
![]()
where, is tangential stress in the cladding respectively; P293 is pressure under the cladding at room temperature, Dmid and t – average cladding diameter and wall thickness respectively, T – cladding temperature (in К).
It should be noted that in these tests the stressed condition of the dummy cladding changes with temperature according to the Charles law, just like on the fuel rod cladding during handling and long-term storage.
Changes in tangential stresses in the dummy claddings under pressure of 3, 4, and 5 MPa during tests in the mode simulating fuel handling and normal operating conditions are shown in Fig. 3.
Changes simulating the handling temperature mode and the limiting design-basis accidents included:
— Fuel rod dummy tests under pressure of 3, 4 and 5 MPa in the mode: heating to 450 C, exposure at 450 C for 3 hours, cooling to 300 C; exposure at 300 C for 1 hour and subsequent three thermal cycles from 300 C (temperature after 2 years of storage) to 450 С. Exposure in each of the cycles at 300 and 450 C for 1 hour (Mode 2) (Fig. 4).

Fig. 4 – Test Temperature Mode with Three Thermal Cycles 300450С. Mode 2
— Fuel rod dummy tests under pressure of 5 MPa in the mode: heating to 450 C, exposure at 450 C for 3 hours, cooling to 300 C; exposure at 300 C for 1 hour and subsequent five thermal cycles from 300 C to 450 С. Exposure in each of the cycles at 300 and 450 C for 1 hour (Mode 3).
— Fuel rod dummy tests under pressure of 5 MPa in the mode: heating to 450 C, exposure at 450 C for 3 hours, cooling to 300 C and subsequent seven thermal cycles from 300 to 450 С. Exposure in each of the cycles for 1 hour at 300 C and 45 minutes at 450 C (Mode 4).
— Fuel rod dummy tests under pressure of 4 and 5 MPa in the mode: heating to 450 C, exposure at 450 C for 3 hours, cooling to 180C and subsequent three thermal cycles from 180 C (temperature after 45-50 years of storage) to 450 С. Exposure in each of the cycles at 180 and 450 C for 1 hour (Mode 5).
— Fuel rod dummy tests under pressure of 4 and 5 MPa in the mode: heating to 450 C, exposure at 450 C for 3 hours, cooling to 180 C and subsequent five thermal cycles from 180 C to 450 C. Exposure in each of the cycles at 180 and 450 C for 1 hour (Mode 6).
— Fuel rod dummy tests under pressure of 5 MPa in the mode: heating to 450 C, exposure at 450 C for 3 hours, cooling to 180 C and subsequent seven thermal cycles from 180 C to 450 C. Exposure in each of the cycles for 1 hour at 180 C and 45 minutes at 450 C (Mode 7).
Metallographic Examination
The metallographic examinations were done using the optical microscope “Axio Observer.A1m”. The metallographic structure images were used to determine the hydrogen concentration and the hydride orientation coefficient.
The hydrogen concentration was determined by the area taken by hydrides in the sample transverse cross-section [22, 23]:

Results and discussion
Re-orientation of Hydrides
During the tests, depending on the concentration, the hydrides which initially had a technology-induced orientation relative to the main directions in products (for tubes: radially, tangentially, and axially) dissolve completely or partially, whereas the subsequent cooling releases hydrides whose orientation is to a significant degree determined by the active stresses. Hysteresis exists between the temperature of full hydride dissolution (TSSD) during heatup and the temperature of their precipitation during cooling (hydride precipitation requires some subcooling). The hydrogen concentrations at hydride dissociation temperatures ([H]TSSD) during heatup and at the beginning of hydride precipitation during cooling ([H]TSSP) in Zr-1%Nb alloy (M5) are determined by the equations [24]:

where: R – gas constant (8.314 J/(moleK); T – absolute temperature, K.
The dependence of hydride dissociation temperatures in Zr-1%Nb during heat up (TSSD) and their precipitation during cooling (TSSP) are shown in Fig. 5.

Fig. 5 – Hydride dissociation temperatures (TSSD) during heat up and at the hydride beginning of precipitation temperatures during cooling (TSSP) in Zr-1%Nb versus hydrogen concentration [H]
The hydrogen reorientation tests in the dummy claddings under pressure of 3, 4, and 5 MPa (at room temperature) simulating fuel handling and normal storage conditions in the SNF dry storage was performed on the dummies with the hydrogen concentration in Zr-1%Nb cladding of 170, 250, 320, and 400 ppm. Fig. 9 marks the hydride dissolution and precipitation temperatures at these concentrations (I-V), as well as the maximum test temperature (450 C (723 K)). The hydride distribution in the dummy claddings before and after the tests is shown in Fig. 6; the data on hydride orientation (hydride orientation coefficients in the dummy claddings) is provided in Table 2.
 |
 |
| 190 ppm, before test, Fn0.04 | 170 ppm, Р293=3.0 MPa, σ at 450С=46 MPa, Fn0.092 |
 |
 |
| 300 ppm, before test, Fn0.068 | 300 ppm, Р293=4.0 MPa, σ at 450С=61.3 MPa, Fn0.58 |
 |
 |
| 400 ppm, before test, Fn0.072 | 400 ppm, Р293=5.0 MPa, σ at 450С=76,7 MPa, Fn0.82 |
Fig. 6 – Hydride microstructure in the dummy (Zr-1%Nb) claddings before and after the hydride reorientation tests in the mode simulating fuel handling in the SNF dry storage (on the right)
Table 2 – Hydride reorientation coefficients in the dummy claddings tests in the mode simulating fuel handling in the SNF dry storage (single cycle heating to 450 °C, holding for 8 hours and subsequent cooling to room temperature (according to regime 1)).

The results obtained (Fig. 6, Table 2) evidence that the dummy tests under the pressure of 3, 4, and 5 MPa in the mode simulating fuel handling and normal storage conditions do not lead to hydride reorientation in the claddings with low hydrogen concentration (60 and 170 ppm (Fig. 5)), whose hydride dissociation and precipitation temperatures are well below 450 C. This may be related to low stresses in the dummy claddings at the temperature of precipitation onset of low-hydrogen concentration hydrides. At hydrogen concentrations of 250…400 ppm, with a pressure increase from 0 to 4 MPa, the hydride orientation coefficient increases only insignificantly, but rises dramatically with a pressure increase from 4 to 5 MPa (Fig. 6, Table 2). Zr-1%Nb cladding propensity towards radial hydride formation under internal pressure increases with the hydrogen concentration growing from 250 to 400 ppm, which is in good qualitative agreement with the dependence of tangential stress in the dummy claddings at the hydride precipitation temperature (TSSP) (See Fig. 5).
For illustration, Fig. 7 shows the dependence of the hydride orientation coefficient versus tangential stress at 450 C for the dummy claddings (Zr-1%Nb) with a hydrogen concentration of 400 ppm (TSSP=451.96 C), tested in the mode simulating fuel handling and normal storage conditions in the SNF Dry Storage, as well as in the modes with three, five, and seven thermal cycles 300-450 C.
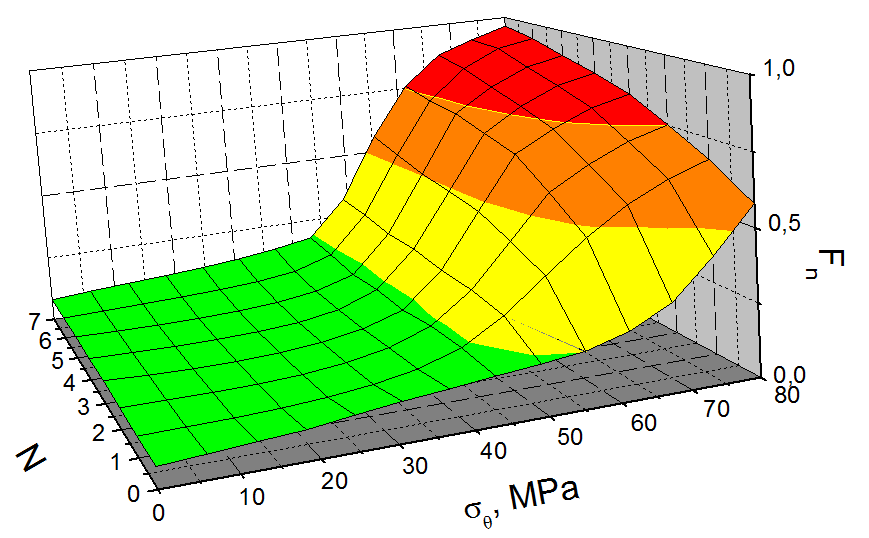

 |
 |
|
|
 |
 |
|
|
Fig. 8 — Hydride microstructure in the dummy claddings (Zr-1%Nb) before and after the hydride reorientation test in the mode: heating to 450 C, exposure for 3 hours, and cooling to 300 С and subsequent 3 (a,b), 5 (c), and 7 (d) thermal cycles 300-450 C


Three thermal cycles of 300-450 C significantly increase the hydride reorientation level, with significant reorientation taking place in the dummy claddings with a hydrogen concentration of 150 ppm, whereas in the dummy claddings with a hydrogen concentration of 250…400 ppm significant reorientation also takes place during pressurized tests at Р293=4 MPa.
It should be noted that the hydride orientation coefficient is significantly increased by three thermal cycles of 300-450 C, whereas further increase in the number of cycles to 5 and 7 (P293=5 MPa) causes the hydride orientation coefficient to grow only insignificantly (see Table 2 and 3; Fig. 7).
Hydride distribution in the dummy claddings before and after the tests in the modes: heating to 450 C, exposure for 3 hours, cooling to 180 C and subsequent 3, 5, and 7 thermal cycles of 180-450 C is shown in Fig. 9; the data on hydride orientation (hydride orientation coefficients in the dummy claddings) is provided in Table 4.
 |
 |
|
|
 |
 |
|
|
Fig. 9 — Hydride microstructure in the dummy claddings (Zr-1%Nb) before and after the hydride reorientation test in the mode: heating to 450 C, exposure for 3 hours, and cooling to 180 С and subsequent 3 (a,b), 5 (c), and 7 (d) thermal cycles 180-450C
Table 4 — Hydride reorientation coefficient in the dummy claddings tested in the mode: heating to 450 C, exposure for 3 hours, cooling to 180С, and subsequent 3, 5, and 7 thermal cycles 180-450 C



As in the case of three 300-450 C thermal cycles, three 180-450 C thermal cycles substantially increase the hydride reorientation level, with significant reorientation taking place in the dummy claddings with a hydrogen concentration of 170 ppm, whereas in the dummy claddings with a hydrogen concentration of 250…400 ppm significant reorientation also takes place during pressurized tests at Р293=4 MPa. The hydride orientation coefficient is significantly increased by three 180-450 C thermal cycles, whereas the effects of further increase in the number of cycles to 5 and 7 (P293=5 MPa) is insignificant and ambiguous (see Table 4).


а) 20 C |

b) 350 C |
Fig. 10 – Dependence of the strength limit, yield strength, and relative elongation of Zr-1% Nb (E110) cladding tubes versus hydrogen concentration. Test temperatures: 20 and 350 C
No temperature mode used in the work led to any additional hydrogen embrittlement during the hydride reorientation tests. For illustration, Fig. 11a and 11b provide dependencies of the strength limit, yield strength, and relative elongation of the samples cut out from dummy claddings under pressure (P293=5 MPa) after 7 thermal cycles 300-450 C, which caused virtually complete hydride reorientation (Fn=0.98), versus hydrogen concentration. After testing in this mode at room temperature, the strength of all the hydrogenated dummy claddings is slightly higher than of the non-hydrogenated ones, and their plasticity virtually does not decrease below 30%. At 350 C the strength of the dummy claddings virtually does not depend on the hydrogen concentration and their plasticity is slightly higher on the hydrogenated dummy claddings.

а) 20 C |

b) 350 C |
Fig. 11 – Dependence of the strength limit, yield strength, and relative elongation of the Zr-1% Nb (E110) dummy claddings tested with 7 thermal cycles 300-450 C; Test temperatures: 20 and 350 C


Reference
[1] Займовский, А. С., Никулина, А. В., and Решетников, Н. Г., “Циркониевые сплавы в ядерной энергетике,” Москва: Энергоатомиздат, 1994. [2] Machiels, A., “End-of-Life Rod Internal Pressures in Spent Pressurized Water Reactor Fuel,” EPRI Product ID: 3002001949 EPRI, California, Palo Alto: Electric Power Research Institute, 2013. [3] Cappelaere, C., Limon, R., Gilbon, D., Bredel, T., Rabouille, O., Bouffioux, P., and Mardon, J. P., “Impact of Irradiation Defects Annealing on Long-Term Thermal Creep of Irradiated Zircaloy-4 Cladding Tube,” Zirconium in the Nuclear Industry: Thirteenth International Symposium, ASTM STP 1423, G. D. Moan and P. Rudling, Eds., ASTM International, West Conshohocken, PA, pp. 720-739, 2002. [4] Couet, A., Motta, A. T., Comstock, R. J., “Hydrogen pickup measurements in zirconium alloys: Relation to oxidation kinetics,” Journal of Nuclear Materials, Vol. 451, № 1-3, pp. 1–13, 2014. [5] Coleman, C. E., and Hardie, D., “The Hydrogen Embrittlement of -Zirconium – A Review,” Journals of the Less Common Metals, Vol. 11, № 3, pp. 168-185, 1966. [6] Bertolino, G., Meyer, G., Perez Ipina, J. “Degradation of the mechanical properties of Zircaloy-4 due to hydrogen embrittlement,” Journal of Alloys and Compounds, Vol. 330–332, pp. 408–413, 2002. [7] Nuttall, K, and Rogowski, A. J., “Some Fractographic aspects of Hydrogen-Induced Delay Cracking in Zr-2.5%Nb alloys,” Journals of Nuclear Materials, Vol. 80, № 2, pp. 279-290, 1979. [8] IAEA-TECDOC-1649, “Delayed Hydride Cracking of Zirconium Alloy Fuel Cladding,” Vienna: IAEA, 2010, ISSN 1011-4289. [9] Coleman, C. , Grigoriev, V., Inozemtsev, V., Markelov, V., Roth, M., Makarevicius, V., Kim, Y. S., Liagat Ali, K., Chakravartty, J.K., Mizrahi, R., and Lalgudi, R, “Delayed Hydride Cracking in Zircaloy Fuel Cladding – An IAEA Coordinated Research Programme,” Nuclear Engineering and Technology, Vol. 41, № 2, pp. 171-178, March 2009. [10] Raynaud, P. A., Koss, D. A., and Motta, A. T., «Crack growth in the through-thickness direction of hydrided thin-wall Zircaloy sheet,» Journal of Nuclear Materials, Vol. 420, № 1-3, pp. 69-82, 2012. [11] “Review Of Used Nuclear Fuel Storage and Transportation. Technical Gap Analyses,” FCRD-USED-2012-000215 Draft, PNNL-21596, Prepared for the U.S. Department of Energy Used Fuel Disposition Campaign, Washington, D.C., July 31 2012. [12] Louthan, M. R., Jr., and Marshall, R. P., “Control of Hydride Orientation of Zircaloy,” Journal of Nuclear Materials, Vol. 9, № 2, pp. 170-184, 1963. [13] Hardie, D., and Shanaham, A. W., “Stress Reorientation of Hydrides in Zirconium-2.5%Nb,” Journal of Nuclear Materials, Vol. 55, № 1, pp. 1-13, 1975. [14] Billone, M. C., Burtseva, T. A., and Einziger, R. E., “Ductile-to-Brittle Transition Temperature for High-Burnup Cladding Alloys Exposed to Simulated Drying-Storage Conditions,” Journal of Nuclear Materials, Vol. 433, № 1-3, pp. 431-448, 2013. [15] Cinbiz, M. N., Koss, D. A., Motta, A. T., ”The Effect of Stress Biaxiality on Hydride Reorientation Threshold Stress,” Proceedings of LWR Fuel Performance Meeting, TopFuel 2015, 13 — 17 September, 2015, Zurich, Switzerland; Belgium, Brussels, Part II, pp. 94-103, 2015. [16] ISG 15, Interim Staff Guidance – 15. Materials Evaluation. [17] NUREG 1536, “Standard Review Plan for Spent Fuel Dry Storage Systems at a General License Facility”, Division of Spent Fuel Storage and Transportation, Office of Nuclear Material Safety and Safeguards, 2009. [18] Новоселов, А. Е., Павлов, С. В., Поленок, В. С., Марков, Д. В., Жителев, В. А., Кобылянский, Г. П., Костюченко, А. Н., и Волкова, И. Н., “Состояние оболочек ТВЭЛов ВВЭР после шести лет эксплуатации,” Физика и химия обработки материалов, № 2, pp. 24-32, 2009. [19] IAEA-TECDOC-1381, “Analysis of differences in fuel safety criteria for WWER and western PWR nuclear power plants,” IAEA, Vienna, 2003. [20] Shebaldov, P. V., Peregud, M. M., Nikulina, A. V., Bibilashvili, Y. K., Lositski, A. F., Kuz’menko, N. V., Belov, V. I., and Novoselov, A. E., “E110 alloy cladding tube properties and their interrelation with alloy structure-phase condition and impurity content,” Zirconium in the Nuclear Industry, Twelfth International Symposium, ASTM STP 1354, G. P. Sabol and G. D. Moan, Eds., American Society for Testing and Materials, West Conshohocken, PA, pp. 545-559, 2000. [21] Лучна, А. Є., Лавренчук, A. І., Сєднєв, В. А., Васильченко, В. М., Двоєглазов, О. М., Медведєв, В. І. и Печера, Ю. М., “Сухе сховище відпрацьаного ядерного палива запорізької АЕС. Забезпечення безпеки,” International Conference Current Problems in Nuclear Physics and Atomic Energy, May 29 – June 3, Kyiv, Ukraine, 2006. [http://www.kinr.kiev.ua/NPAE_Kyiv2006/proc/Luchna.pdf]. [22] Daum, R. S., Chu, Y. S., and Motta, A. T., “Identification and quantification of hydride phases in Zircaloy-4 cladding using synchrotron X-ray diffraction,” Journal of Nuclear Materials, Vol. 392, № 3, pp. 453-464, 2009. [23] Liu, Y., Peng, Q., Zhao, W., and Jiang, H., “Hydride precipitation by cathodic hydrogen charging method in zirconium alloys,” Materials Chemistry and Physics, Vol. 110, № 1, pp. 56-60, 2008. [24] Tang, R., and Yang, X., “Dissolution and precipitation behaviors of hydrides in N18, Zry-4 and M5 alloys,” International Journal of Hydrogen Energy, Vol. 34, № 17, pp. 7269–7274, 2009.